Comparison of measurement methods for concentration and density measurement
Each of these measurement methods has advantages and disadvantages compared to the others. Click on the respective measurement method.
Sound velocity measurement
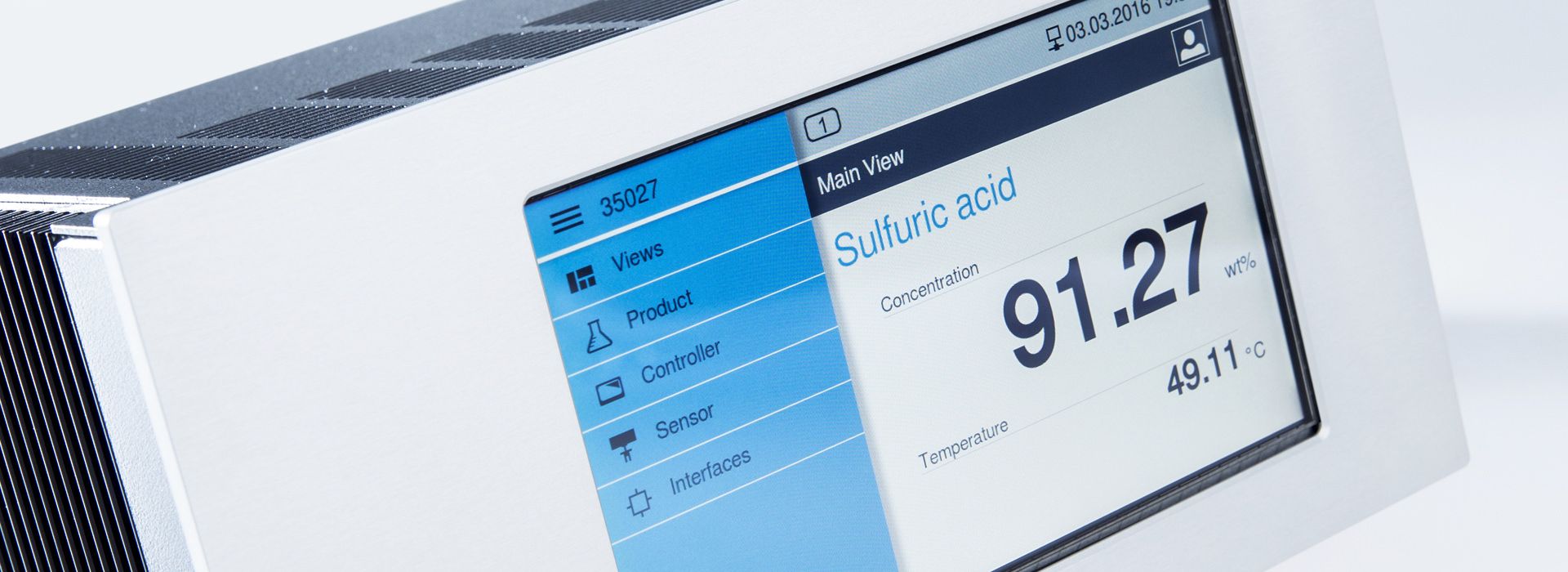
The LiquiSonic® technology is based on sound velocity measurement. This ultrasonic measurement method allows precise and quick determination of the concentration in a liquid, as the sound velocity in a liquid depends on the concentration of the individual components and the temperature.
To determine the sound velocity, a sound pulse is sent through the liquid and the time is measured until the pulse reaches the receiver. Since the distance between the ultrasonic transmitter and receiver is constant by design, the sound velocity can be calculated.
Properties of the LiquiSonic® measurement method:
- Measurement is independent of the color, conductivity, and transparency of the process liquid
- Measurement accuracy of ±0.05 m/s
- High, drift-free measurement accuracy even with a high gas bubble content
- Measurement update rate of 250 ms
- Installation directly in pipes or containers
- Bypass-free installation
- Robust sensor design without seals or moving parts
- Maintenance-free measurement technology
- Insensitive to contamination
- Operating temperature from –90 °C to 200 °C
- Operating pressure of up to 500 bar
- Use of special materials in chemically aggressive liquids
- Integrated temperature measurement
Coriolis density measurement
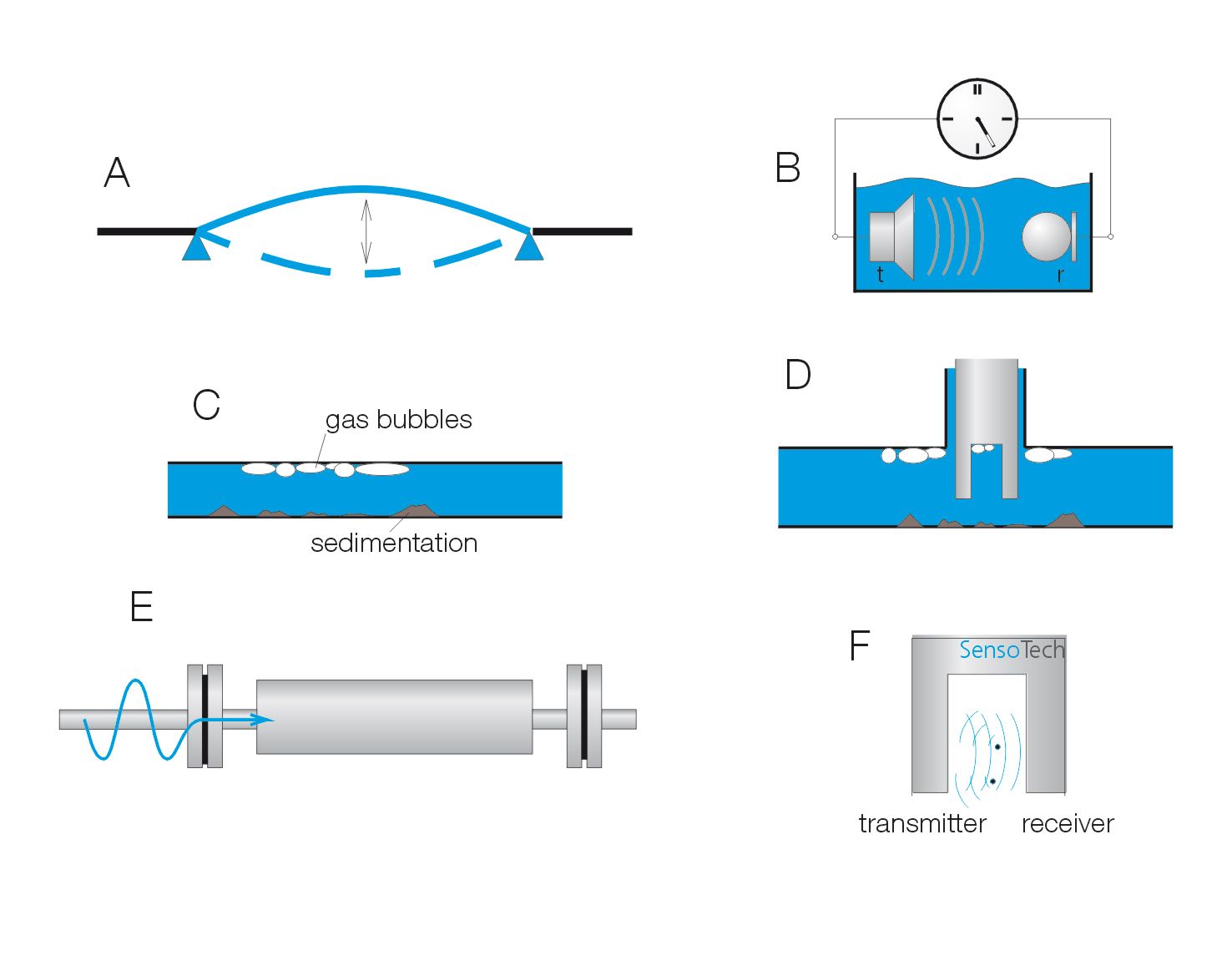
The phase shift of a flowing, vibrating tube is related to the mass flow of the flowing liquid via the Coriolis force. Various manufacturers of Coriolis mass flow meters promote the suitability of their devices for determining density.
However, Coriolis flow meters are structurally optimized for detecting the phase shift of the oscillation system, while density measurement requires precise measurement of the oscillation frequency. Therefore, the achievable accuracy is in most cases worse than ± 5% to ± 10% of the measuring range.
Density and concentration measurement is therefore only conditionally possible and associated with many disadvantages:
- high sensitivity to gas bubbles and sediments
- implementation of a temperature compensation for the device, but not for the density calculation
- only factory density calibration possible
- high installation effort for larger nominal widths
- internal reduction of nominal width, hence high pressure drop and susceptibility to contamination
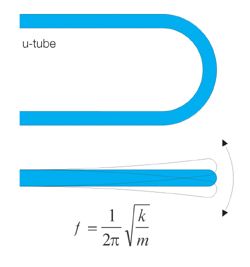
bending oscillator density measurement
The bending oscillator principle is a proven method in the laboratory field for density measurement and uses the dependence of the oscillation frequency of a flowing tube on the density of the flowing liquid.
However, in process applications, this method encounters the following limitations:
- usable only in bypass, maximum nominal width typically 10 mm
- the bending oscillator is pressure-sensitive and shock-sensitive
- no immersion sensors feasible
- high sensitivity to gas bubbles and sediments
conductivity measurement
The conductivity of a liquid depends on the concentration and activity of the electrically conductive ions in that liquid.
The (inductive) conductivity measurement is therefore an inexpensive method for concentration measurement, but it has the following disadvantages:
- The activity and thus the conductivity of the ions is highly temperature-dependent (up to 3% per °C).
- The activity is strongly influenced by impurities, complex formations, hydration shells, etc.
- The method is inherently applicable only for measurement tasks in inorganic liquids.
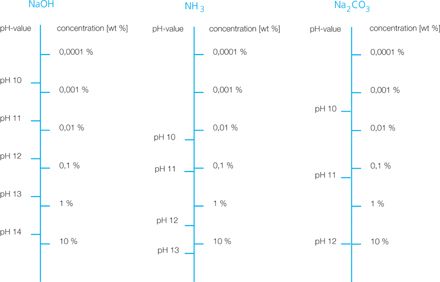
pH value measurement
The determination of the pH value is a method adopted from the laboratory for the indirect determination of concentration or density.
However, the advantage of the low price of the sensors used is offset by a number of disadvantages:
- direct contact of the membrane with the process necessary
- high drift requires ongoing calibration effort as well as complex and expensive fittings and sampling technology
- not usable in typical concentration measurement ranges of greater than 1 m%
- pH sensors are made of glass; due to their fragility, their use is critical in certain industries (food, pharmaceuticals)
Refractometry
Determining the critical angle of total reflection (refractive index) is a laboratory method used to determine concentration or density through calibration curves.
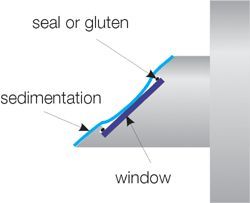
The refractive index is determined at the optical window. This results in a number of disadvantages for process devices (refractometers):
- Deposits on the window cause a drift in measurement values or prevent measurement.
- Optical windows require a seal or adhesive that can be attacked by corrosive process fluids.
- Parts of the electronics (CCD line) require Peltier cooling, resulting in a limited lifespan.
- The refractive index depends on the wavelength of the light.
- Refractive index values from literature or handheld/laboratory refractometers cannot be used for process devices.
Radiometry
A radioactive source emits radiation onto the measurement material, which is received by the detector. A scintillator converts the radioactive radiation into light flashes and evaluates their number. Since the penetration of gamma radiation depends on the material, the density is determined from the intensity of the incoming radiation.
1: Source with shielding
2: Scintillation counter
3: Clamp-on measurement section on the pipeline
Radiometry is being replaced by modern measurement methods today because the use of radiometric measurement involves high effort, regulatory requirements, costs, and potential hazards:
- Complex, expensive approval of devices by TÜV / professional associations
- Ongoing maintenance effort, e.g., regular leak tests
- Training of radiation protection officers
- Information and documentation obligations towards the fire department
- Very expensive disposal of radiation sources in case of replacement or return of devices
- Delivery in special vehicles
- High risk potential for employees in case of accidents
The concentration of a liquid can be determined using various measurement methods.
A concrete comparison between LiquiSonic® and alternative measurement methods can be downloaded below.
Concentration measurement in liquids
Comparison of measurement methods